New observational study launched to assess PML survivors
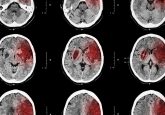
Inhibikase Therapeutics Inc. have today announced the launch of an observational clinical study in patients with progressive multifocal leukoencephalopathy (PML), which is a possible side effect of a variety of drugs used in treatment for diseases such as multiple sclerosis, Crohn’s disease, ulcerative colitis and hematologic cancers as well as HIV/AIDS patients. JC Virus (JCV) is the primary cause of PML and its transport from the kidney into the brain can be induced by a range of medications. Once in the brain JCV is capable of causing severe and irreversible damage which can lead to death. “The company has a...