Organoids: grow your own brain
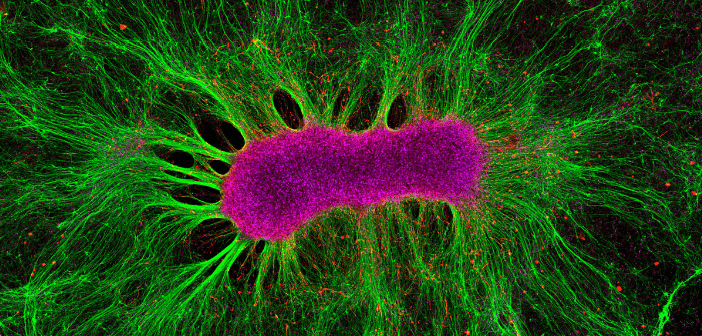
Credit: Collin Edington and Iris Lee, Koch Institute MIT. CC BY-NC-ND
Jenny Straiton explores the miniature world of organoids and discusses how these small models are making big changes in the world of neurological research. Initially, scientists learned about disorders when disaster happened – Paul Broca made his mark when he linked posterior left frontal lobe damage to speech production; losing his hippocampus made Henry Molaison, better known as H.M., the world's most famous amnesiac; and being run through with a railroad spike turned Phineas Gage into a psychological phenomenon. Later, neuroscientists relied on in vitro cell cultures or animal models to see what was happening. Although less devastating for the...